
Testing the preservation potential of early diagenetic dolomites as geochemical archives - Mueller - 2020 - Sedimentology - Wiley Online Library
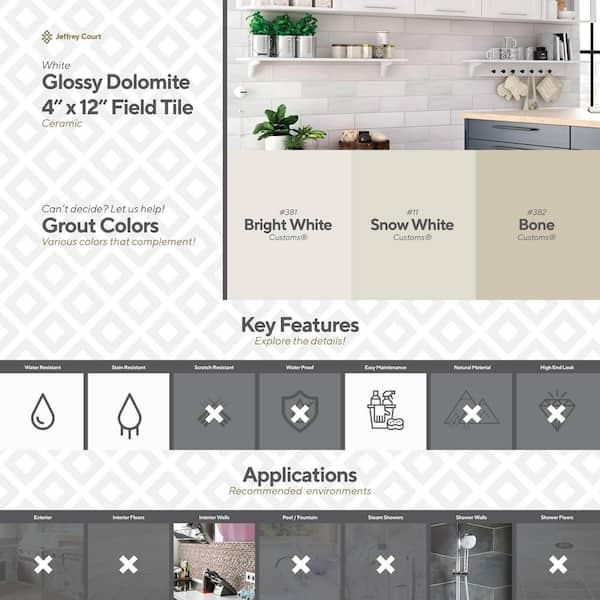
Jeffrey Court Glossy Dolomite White 4 in. x 12 in. Subway Gloss Ceramic Wall Tile (9.687 sq. ft./Case) 93024 - The Home Depot

Lucida Surfaces MaxCore Dolomite 1-3/4 in. W x 47-1/4 in. L x 0.27 in. T Vinyl T-Molding MC-506TM - The Home Depot

Mechanical data and microstructures of deformed room-humidity dolomite... | Download Scientific Diagram






















