
Mineralogy, nucleation and growth of dolomite in the laboratory and sedimentary environment: A review - Gregg - 2015 - Sedimentology - Wiley Online Library

Ruminal Solubility and Effect of Calcite Powder Supplementation on Dairy Animal Performance | Semantic Scholar
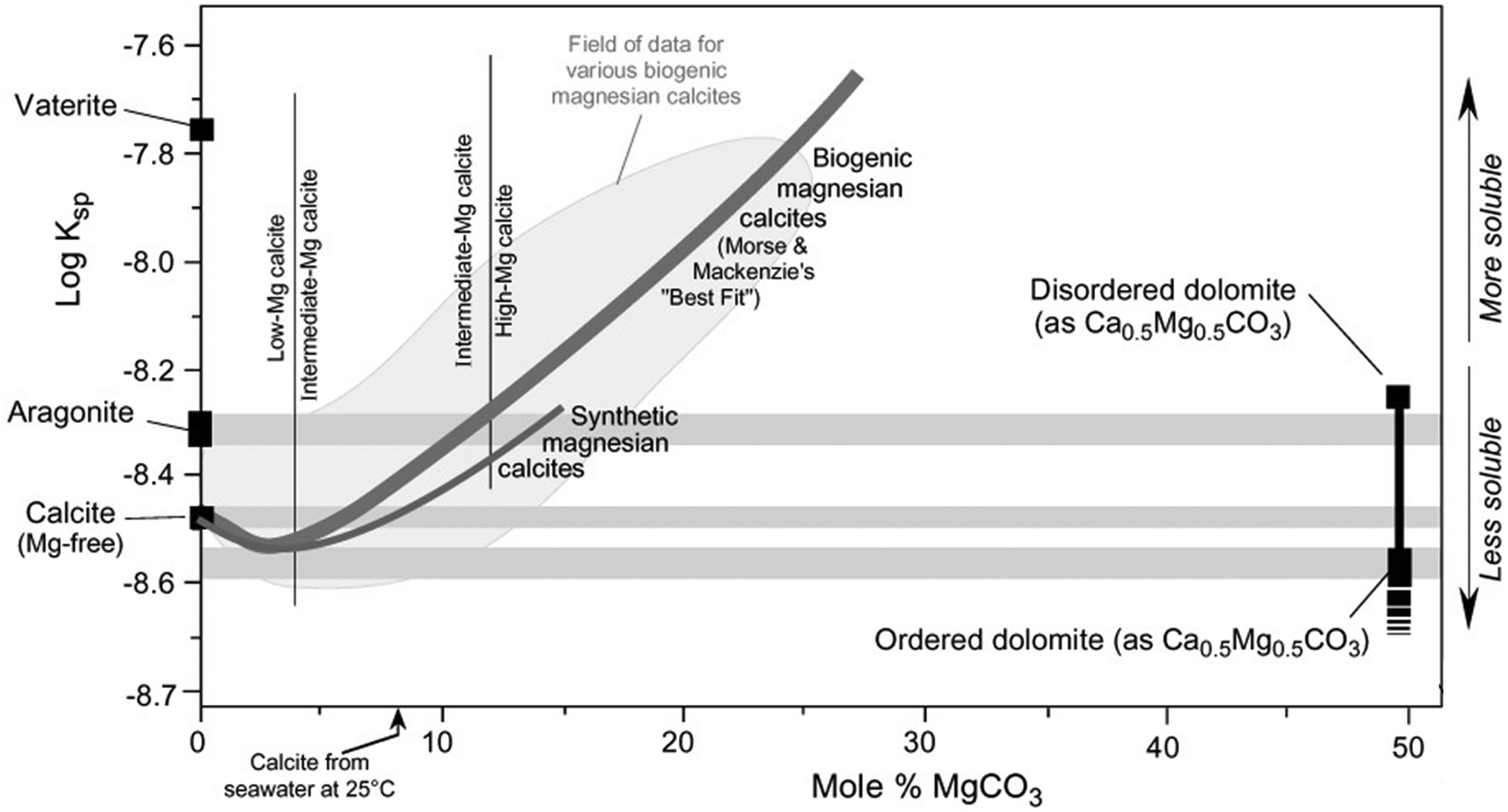
Mechanisms of Mg carbonates precipitation and implications for CO 2 capture and utilization/storage - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D2QI02482A
Self-accelerating volumetric dolomite-for-calcite replacement: A possible mechanism for high-temperature dolomitization? | SpringerLink

On the interaction between calcite and dolomite: Insights from gas and aqueous geochemistry and mineralogical characterization - ScienceDirect

Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150 °C and 1 to 55 atm pCO2: New constraints on CO2 sequestration in sedimentary basins - ScienceDirect

5 Temperature Control of Mineral Deposition – A Conceptual Overview of Surface and Near Surface Brines and Evaporite Minerals

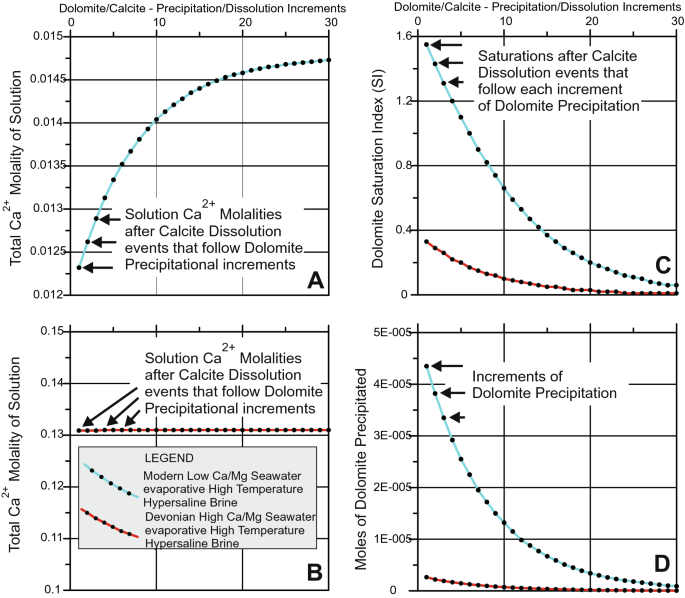
The impact of Mg2+ ions on equilibration of Mg-Ca carbonates in groundwater and brines - ScienceDirect













